Increasing the efficiency of light-to-electricity conversion has long been a goal of photodetector designers. These micron-sized devices are used in an array of consumer and industrial products, from cell phones to solar cells to the panels on space shuttles.
A design that doubles photodetector efficiency has been engineered at the University of California, Riverside. The increase was achieved by stacking two atomic layers of tungsten diselenide (WSe2) on a single atomic layer of molybdenum diselenide (MoSe2). Different properties than those of the parent layers are realized with this arrangement, allowing for customized electronic engineering at the tiniest possible scale.
How does this increase conversion efficiency? A photon striking the WSe2 layer knocks loose an electron, freeing it to conduct through the WSe2. At the junction between WSe2 and MoSe2, the electron drops down into MoSe2, and the energy given off then bumps a second electron from the WSe2 into the MoSe2, where both electrons become free to move and generate electricity.
“We are seeing a new phenomenon occurring,” said Nathaniel M. Gabor, an assistant professor of physics, 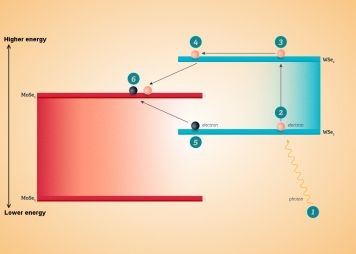 Energy diagram of the WSe2-MoSe2 device. Source: University Communications, UC Riversidewho led the research team. “Normally, when an electron jumps between energy states, it wastes energy. In our experiment, the waste energy instead creates another electron, doubling its efficiency. Understanding such processes, together with improved designs that push beyond the theoretical efficiency limits, will have a broad significance with regard to designing new ultra-efficient photovoltaic devices.”
Energy diagram of the WSe2-MoSe2 device. Source: University Communications, UC Riversidewho led the research team. “Normally, when an electron jumps between energy states, it wastes energy. In our experiment, the waste energy instead creates another electron, doubling its efficiency. Understanding such processes, together with improved designs that push beyond the theoretical efficiency limits, will have a broad significance with regard to designing new ultra-efficient photovoltaic devices.”
In existing solar panels models, one photon can at most generate one electron but in the prototype, one photon can generate two electrons or more through electron multiplication.
The researchers explain that the process of generating two electrons from one photon is perfectly allowable at extremely small length scales. As a material is thinned down to dimensions nearing the electron’s wavelength, the material’s properties begin to change in ways not fully understood.
Increasing the temperature of the device also yields more electrons: a doubling of electrons was observed at 340 degrees Kelvin (150 F).
Electron multiplication in conventional photocell devices typically requires applied voltages of 10 to 100 volts. The researchers used only 1.2 volts, the typical voltage supplied by a AA battery, to achieve a doubling of electrons.
“These materials, being only an atom thick, are nearly transparent,” Gabor said. “It’s conceivable that one day we might see them included in paint or in solar cells incorporated into windows. Because these materials are flexible, we can envision their application in wearable photovoltaics, with the materials being integrated into the fabric. We could have, say, a suit that generates power—energy-harvesting technology that would be essentially invisible.”

