 More molecular design strategies for the creation of light-emitting materials are made possible by the mechanism illustrated here. (Source: William J. Potscavage, Jr.)Organic light-emitting diodes, or OLEDs, have a wealth of applications in next-generation displays and lighting. They produce vibrant colors and can be fabricated into everything — from tiny pixels to large, flexible panels. They work by injecting electrical charges into thin films of organic molecules, producing packets of energy that emit light. These energy packets are known as “excitons.”
More molecular design strategies for the creation of light-emitting materials are made possible by the mechanism illustrated here. (Source: William J. Potscavage, Jr.)Organic light-emitting diodes, or OLEDs, have a wealth of applications in next-generation displays and lighting. They produce vibrant colors and can be fabricated into everything — from tiny pixels to large, flexible panels. They work by injecting electrical charges into thin films of organic molecules, producing packets of energy that emit light. These energy packets are known as “excitons.”
But OLEDs are limited by the fact that three-quarters of these excitons are “triplets,” which do not produce light in conventional materials. The “singlets” representing the remaining one quarter emit light through fluorescence.
There are a few workarounds. Including a rare metal, such as iridium or platinum, in the organic molecule enables those triplets to emit light through phosphorescence. This method involves more cost, and reduces the freedom of molecular design. Another option is to add heat to the environment to boost those triplets into singlets, a process known as thermally activated delayed fluorescence (TADF).
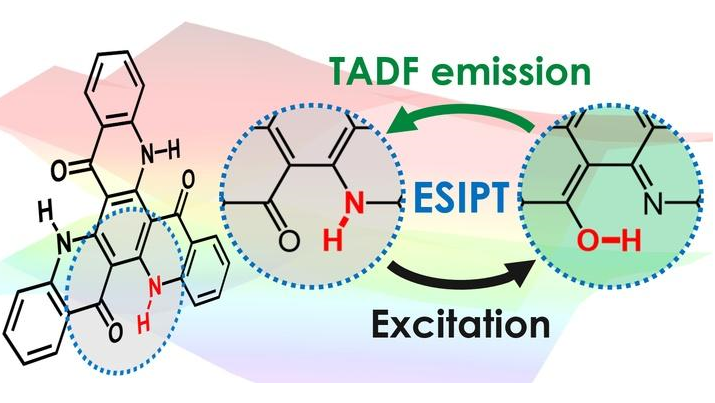
TADF is effective, but scientists want to expand their strategies for incorporating it. That’s why researchers at Kyushu University's Center for Organic Photonics and Electronics Research (OPERA) have taken a new look at a molecule originally synthesized for creating a unique light-absorbing pigment. The mechanism they studied involves the reversible transfer of a hydrogen atom’s positive nucleus from one atom in the emitting molecule to another in the same molecule. When excited with optical or electrical energy, this transfer — known as excited-state intramolecular proton transfer, or ESIPT — occurs spontaneously, without the donor-acceptor electron scheme that is typically employed.
The researchers demonstrated that a molecule that slightly changes its chemical structure before and after emission can achieve high efficiency in organic light-emitting diodes. Their approach represents a new design strategy that opens the door for achieving TADF with a variety of new chemical structures. Molecules similar to the one studied are known to be highly resistant to degradation, which also has implications for improving the lifetime of OLEDs.

